
SF4 is a covalent (polar covalent) compound because when one nonmetal combines with another nonmetal, it usually forms a covalent compound. Here, S is a nonmetal and F is also a nonmetal. So when they combine, it forms a covalent compound.
Well, now you have got to know that SF4 is a covalent compound, but let me explain the in-depth reason why SF4 is a covalent compound.
If you are a visual learner like me, then here is a short one minute video for you.
Why is SF4 a Covalent compound?
As mentioned above, you can simply remember that when the nonmetal combines with another nonmetal, the bond between them is a covalent bond.
Here in SF4, the S atom is a nonmetal and the F atom is also a nonmetal.

Hence the bond between them is a covalent bond.
Is SF4 polar covalent or nonpolar covalent?
In order to know whether SF4 is a polar covalent molecule or nonpolar covalent molecule, we have to check the electronegativity difference of the combining atoms.
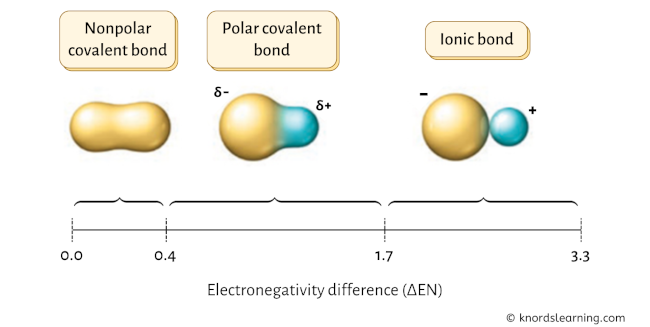
If the electronegativity difference (ΔEN) is less than 0.4, then the bond is nonpolar covalent bond.
If the electronegativity difference (ΔEN) is between 0.4 to 1.7, then the bond is polar covalent bond. [1] [2] [3] [4] [5]
Now the electronegativity of Sulfur and Fluorine are mentioned below. (You can see the electronegativity of all the elements from this electronegativity chart).
- Electronegativity of Sulfur (S) = 2.58
- Electronegativity of Fluorine (F) = 3.98
So for SF4, the electronegativity difference (ΔEN) = 3.98 – 2.58 = 1.4
This value lies between 0.4 to 1.7, which indicates that the bond between Sulfur (S) and Fluorine (F) is polar covalent bond.
But the SF4 molecule has 1 lone pair which results in an asymmetric shape of the entire SF4 molecule.
Because of this asymmetric shape there are positive and negative poles of charges on the overall molecule of SF4.
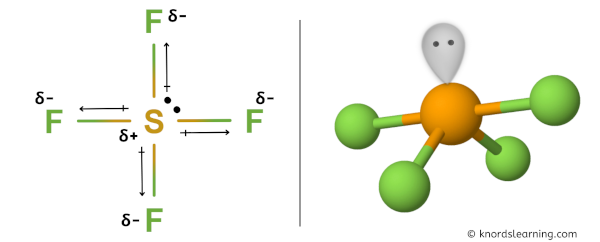
Hence, SF4 is a polar covalent molecule.
I hope you have understood the reason why SF4 is a polar covalent compound.
Check out other compounds to see whether they are ionic or covalent;
Is Br2 Ionic or Covalent?
Is K3PO4 Ionic or Covalent?
Is CsCl Ionic or Covalent?
Is KNO3 Ionic or Covalent?
Is PH3 Ionic or Covalent?
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
