
SO3 is a NONPOLAR molecule.
But why?
And how can you say that SO3 is a nonpolar molecule?
Want to know the reason?
Let’s dive into it!
SO3 is a NONPOLAR molecule because all the three bonds (S=O bonds) are identical and SO3 has symmetrical geometry which cancels out the bond polarity.
Let me explain this in detail with the help of SO3 lewis structure and its 3D geometry.
Why is SO3 a Nonpolar molecule? (Explained in 3 Steps)
SO3 is a nonpolar molecule because it does not have any pole of positive charge and negative charge on it.
Let me explain this to you in 3 steps!
Step #1: Draw the lewis structure
Here is a skeleton of SO3 lewis structure and it contains three S=O bonds.
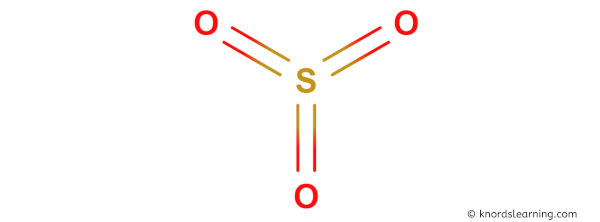
(Note: If you want to know the steps of drawing the SO3 lewis dot structure, then visit this article: SO3 lewis structure, Or you can also watch this short 2 minute video).
So from the above diagram we have come to know that the SO3 molecule has three S=O bonds.
Now in the next step we have to check whether these three S=O bonds are polar or nonpolar.
Step #2: Check whether individual bonds are polar or nonpolar
The chemical bonds can be either nonpolar, polar or ionic depending on the difference of the electronegativity values (ΔEN) between the two atoms.
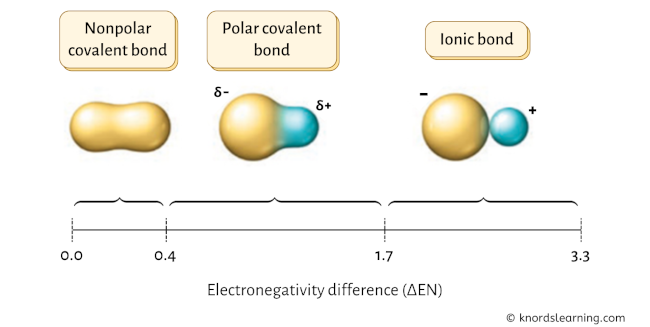
Have a look at the above image.
- If the electronegativity difference (ΔEN) is less than 0.4, then the bond is nonpolar covalent bond.
- If the electronegativity difference (ΔEN) is between 0.4 to 1.7, then the bond is polar covalent bond.
- If the electronegativity difference (ΔEN) is greater than 1.7, then the bond is an ionic bond. [1] [2] [3] [4] [5]
Now let’s come to the example of SO3 molecule. It has three S=O bonds.
You can see the electronegativity values of Sulfur (S) and Oxygen (O) atoms from the periodic table given below.
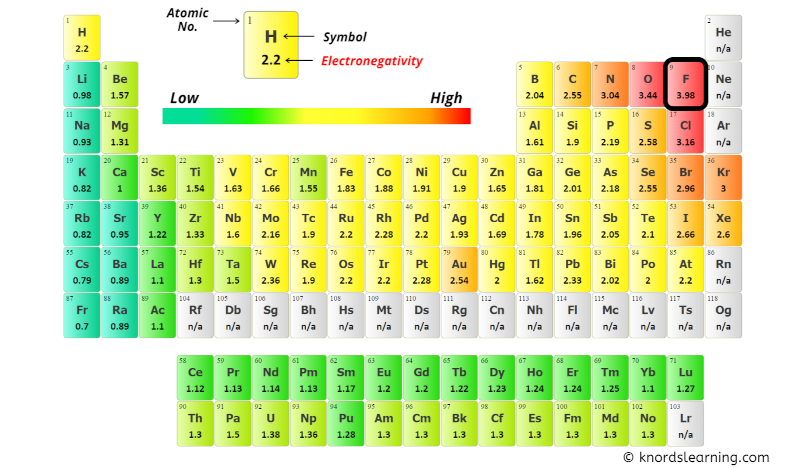
From the above image;
Now let’s see the polarity of each bond.
For Sulfur-Oxygen bond;
The electronegativity difference (ΔEN) = 3.44 – 2.58 = 0.86
This value lies between 0.4 to 1.7, which indicates that the bond between Sulfur (S) and Oxygen (O) is polar.
Hence, each Sulfur-Oxygen bond is a polar covalent bond.
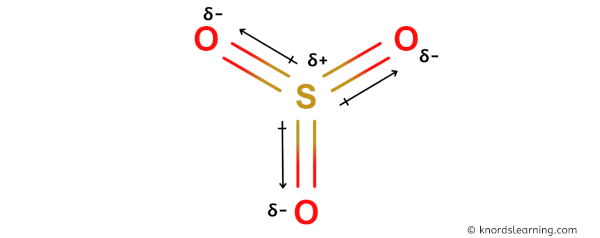
You can see in the above image that because of electronegativity difference, the partial positive charge (ẟ+) appears on the Sulfur atom (S) and partial negative charge (ẟ-) appears on the Oxygen atoms (O).
But wait, this alone won’t tell you whether the entire SO3 molecule is polar or nonpolar.
So let’s proceed to the next step to check the symmetry of the SO3 molecule.
Step #3: Check whether the molecule is symmetric or not
Have a look at this 3D structure of SO3. You can see that the structure of SO3 is symmetrical.
The sulfur atom is at the center and it is surrounded by 3 oxygen atoms which are equidistant as well as at equal angles.
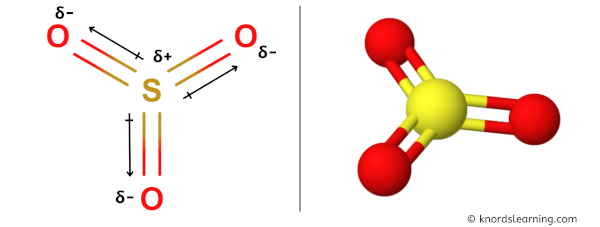
As the bonds (S=O) are symmetrical and the SO3 molecule has a symmetrical geometry, their bond polarity gets canceled with each other.
Because of this, there are no positive and negative poles of charges on the overall molecule of SO3.
Hence, the SO3 molecule is a nonpolar molecule.
I hope you have understood the reason behind the nonpolar nature of SO3 molecule.
See the polarity of other molecules to make your concepts clear:
Is PCl3 Polar or Nonpolar?
Is HCN Polar or Nonpolar?
Is CH2O Polar or Nonpolar?
Is XeF4 Polar or Nonpolar?
Is SF6 Polar or Nonpolar?
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
