
The Charge of N2O (Nitrous oxide) is 0.
But the question is how can you say that the charge on N2O (nitrous oxide) is 0?
Well you can say this by calculating its formal charge.
So let’s calculate the formal charge of N2O (nitrous oxide).
If you are a visual learner like me, then here is a short two minute video for you.
Calculating the formal charge of N2O using lewis structure
In order to calculate the formal charge on N2O (nitrous oxide), you should know the Lewis dot structure of N2O (nitrous oxide).
Here is the lewis structure of N2O.

Now using the above lewis structure of N2O, you have to find the formal charge on each atom that is present in the N2O molecule.
For calculating the formal charge, you need to remember this formula;
Formal charge = Valence electrons – Nonbonding electrons – (Bonding electrons)/2
You can see the bonding and nonbonding electrons of N2O from the image given below.

So now let’s calculate the formal charge on each individual atom present in N2O.
Formal charge on left Nitrogen atom:
Valence electrons = 5 (as it is in group 15 on periodic table) [1]
Nonbonding electrons = 2
Bonding electrons = 6
So according to the formula of formal charge, you will get;
Formal charge on left Nitrogen = Valence electrons – Nonbonding electrons – (Bonding electrons)/2 = 5 – 2 – (6/2) = 0

So the formal charge on left nitrogen atom is 0.
Formal charge on central Nitrogen atom:
Valence electrons = 5 (as it is in group 15 on periodic table)
Nonbonding electrons = 0
Bonding electrons = 8
So according to the formula of formal charge, you will get;
Formal charge on central Nitrogen = Valence electrons – Nonbonding electrons – (Bonding electrons)/2 = 5 – 0 – (8/2) = 1+
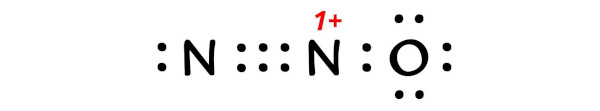
So the formal charge on central nitrogen atom is 1+.
Formal charge on Oxygen atom:
Valence electrons = 6 (as it is in group 16 on periodic table) [2]
Nonbonding electrons = 6
Bonding electrons = 2
So according to the formula of formal charge, you will get;
Formal charge on Oxygen = Valence electrons – Nonbonding electrons – (Bonding electrons)/2 = 6 – 6 – (2/2) = 1-

So the formal charge on oxygen atom is 0.
Now let’s put all these charges on the lewis dot structure of N2O.
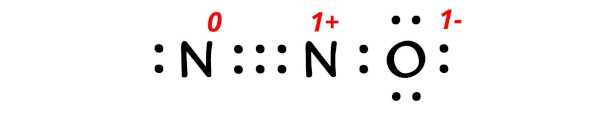
So there is overall 0 charge left on the entire molecule.
This indicates that the N2O (nitrous oxide) has 0 charge.
I hope you have understood the above calculations of N2O (nitrous oxide).
Check out some other related topics for your practice.
Related topics:
Charge of Germanium (Ge)
Charge on H2S
Charge on HBr
Charge on POCl3
Charge on NO3 (Nitrate ion)
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
