
NaOH is an ionic compound because it is formed by two ions, Na+ and OH–. These positive and negative ions produce the force of attraction between them which results in an ionic bond.
Moreover when the metal combines with nonmetal, it usually forms an ionic compound. Here, Na is a metal and OH is a group of nonmetals. So when they combine, it forms an ionic compound.
Well, now you have got to know that NaOH is an ionic compound, but let me explain the in-depth reason why NaOH is an ionic compound.
If you are a visual learner like me, then here is a short one minute video for you.
Why is NaOH an ionic compound?
As mentioned above, you can simply remember that when the metal combines with nonmetal, the bond between them is an ionic bond.
Here in NaOH, the Na atom is a metal and the OH is a group of nonmetals.
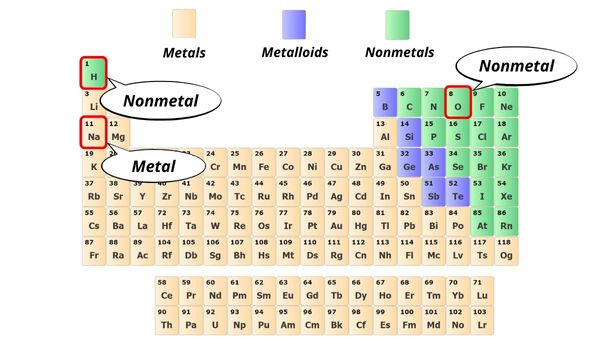
Hence the bond between them is an ionic bond.
But how does the ionic bond form between Na and OH?
When Na and OH combine with each other, the electron transfer takes place from Na to OH.
In other words, Na loses 1 electron and OH gains 1 electron.
Due to this, the Sodium becomes a positive ion Na+ and OH becomes a negative ion OH–.
Now because of the positive charge of Sodium ion and negative charge of OH ion, the electrostatic force of attraction is produced between them.
This electrostatic force between Na ion and OH ion results in an ionic bond between them.
Hence, NaOH is an ionic compound.
I hope you have understood the reason why NaOH is an ionic compound.
Check out other compounds to see whether they are ionic or covalent;
Is HBr Ionic or Covalent?
Is LiF Ionic or Covalent?
Is MgSO4 Ionic or Covalent?
Is LiCl Ionic or Covalent?
Is H2 Ionic or Covalent?
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
