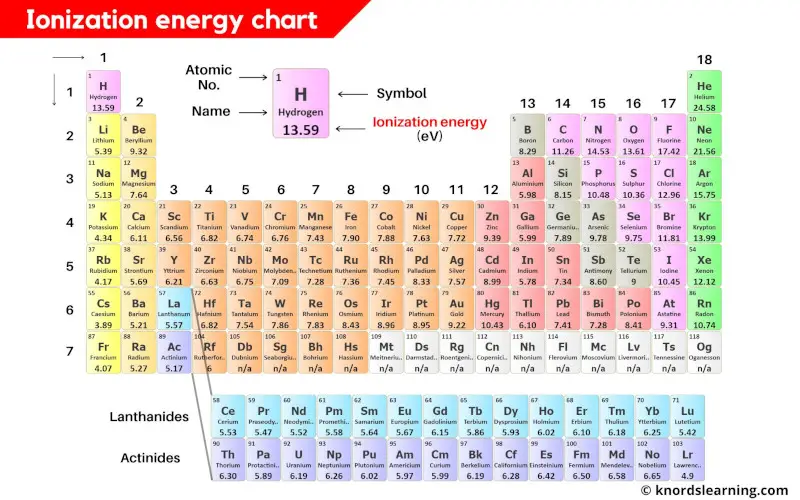
Ionization energy of all the elements of periodic table is mentioned in the above image.
Note: The ionization energy is measured in joules (J) or electron volts (eV). The values mentioned in the above periodic table is the First Ionization energy and are given in electron volts (eV).
The first ionization energy, second ionization energy as well as third ionization energy of the elements are given in this chart below.
Ionization Energy Chart of All Elements of Periodic Table
| Atomic no. | Element | First, Second and Third Ionization Energy (eV) | ||
| First Ionization Energy | Second Ionization Energy | Third Ionization Energy | ||
| 1 | Ionization energy of Hydrogen (H) | 13.59 eV | – | – |
| 2 | Ionization energy of Helium (He) | 24.58 eV | 54.41 eV | – |
| 3 | Ionization energy of Lithium (Li) | 5.39 eV | 75.64 eV | 122.45 eV |
| 4 | Ionization energy of Beryllium (Be) | 9.32 eV | 18.21 eV | 153.89 eV |
| 5 | Ionization energy of Boron (B) | 8.29 eV | 25.15 eV | 37.93 eV |
| 6 | Ionization energy of Carbon (C) | 11.26 eV | 24.38 eV | 47.88 eV |
| 7 | Ionization energy of Nitrogen (N) | 14.53 eV | 29.60 eV | 47.44 eV |
| 8 | Ionization energy of Oxygen (O) | 13.61 eV | 35.11 eV | 54.93 eV |
| 9 | Ionization energy of Fluorine (F) | 17.42 eV | 34.97 eV | 62.70 eV |
| 10 | Ionization energy of Neon (Ne) | 21.56 eV | 40.96 eV | 63.45 eV |
| 11 | Ionization energy of Sodium (Na) | 5.13 eV | 47.28 eV | 71.62 eV |
| 12 | Ionization energy of Magnesium (Mg) | 7.64 eV | 15.03 eV | 80.14 eV |
| 13 | Ionization energy of Aluminum (Al) | 5.98 eV | 18.82 eV | 28.44 eV |
| 14 | Ionization energy of Silicon (Si) | 8.15 eV | 16.34 eV | 33.49 eV |
| 15 | Ionization energy of Phosphorus (P) | 10.48 eV | 19.76 eV | 30.20 eV |
| 16 | Ionization energy of Sulfur (S) | 10.36 eV | 23.33 eV | 34.79 eV |
| 17 | Ionization energy of Chlorine (Cl) | 12.96 eV | 23.81 eV | 39.61 eV |
| 18 | Ionization energy of Argon (Ar) | 15.75 eV | 27.62 eV | 40.74 eV |
| 19 | Ionization energy of Potassium (K) | 4.34 eV | 31.63 eV | 45.80 eV |
| 20 | Ionization energy of Calcium (Ca) | 6.11 eV | 11.87 eV | 50.91 eV |
| 21 | Ionization energy of Scandium (Sc) | 6.56 eV | 12.79 eV | 24.75 eV |
| 22 | Ionization energy of Titanium (Ti) | 6.82 eV | 13.57 eV | 27.49 eV |
| 23 | Ionization energy of Vanadium (V) | 6.74 eV | 14.66 eV | 29.31 eV |
| 24 | Ionization energy of Chromium (Cr) | 6.76 eV | 16.48 eV | 30.96 eV |
| 25 | Ionization energy of Manganese (Mn) | 7.43 eV | 15.63 eV | 33.66 eV |
| 26 | Ionization energy of Iron (Fe) | 7.90 eV | 16.18 eV | 30.65 eV |
| 27 | Ionization energy of Cobalt (Co) | 7.88 eV | 17.08 eV | 33.50 eV |
| 28 | Ionization energy of Nickel (Ni) | 7.63 eV | 18.16 eV | 35.19 eV |
| 29 | Ionization energy of Copper (Cu) | 7.72 eV | 20.29 eV | 36.84 eV |
| 30 | Ionization energy of Zinc (Zn) | 9.39 eV | 17.96 eV | 39.72 eV |
| 31 | Ionization energy of Gallium (Ga) | 5.99 eV | 20.51 eV | 30.71 eV |
| 32 | Ionization energy of Germanium (Ge) | 7.89 eV | 15.93 eV | 34.22 eV |
| 33 | Ionization energy of Arsenic (As) | 9.78 eV | 18.63 eV | 28.35 eV |
| 34 | Ionization energy of Selenium (Se) | 9.75 eV | 21.19 eV | 30.82 eV |
| 35 | Ionization energy of Bromine (Br) | 11.81 eV | 21.8 eV | 36 eV |
| 36 | Ionization energy of Krypton (Kr) | 13.99 eV | 24.35 eV | 36.95 eV |
| 37 | Ionization energy of Rubidium (Rb) | 4.17 eV | 27.28 eV | 40 eV |
| 38 | Ionization energy of Strontium (Sr) | 5.69 eV | 11.03 eV | 42.89 eV |
| 39 | Ionization energy of Yttrium (Y) | 6.21 eV | 12.24 eV | 20.52 eV |
| 40 | Ionization energy of Zirconium (Zr) | 6.63 eV | 13.13 eV | 22.99 eV |
| 41 | Ionization energy of Niobium (Nb) | 6.75 eV | 14.32 eV | 25.04 eV |
| 42 | Ionization energy of Molybdenum (Mo) | 7.09 eV | 16.16 eV | 27.13 eV |
| 43 | Ionization energy of Technetium (Tc) | 7.28 eV | 15.26 eV | 29.54 eV |
| 44 | Ionization energy of Ruthenium (Ru) | 7.36 eV | 16.76 eV | 28.47 eV |
| 45 | Ionization energy of Rhodium (Rh) | 7.45 eV | 18.08 eV | 31.06 eV |
| 46 | Ionization energy of Palladium (Pd) | 8.33 eV | 19.43 eV | 32.93 eV |
| 47 | Ionization energy of Silver (Ag) | 7.57 eV | 21.49 eV | 34.83 eV |
| 48 | Ionization energy of Cadmium (Cd) | 8.99 eV | 16.90 eV | 37.48 eV |
| 49 | Ionization energy of Indium (In) | 5.78 eV | 18.86 eV | 28.03 eV |
| 50 | Ionization energy of Tin (Sn) | 7.34 eV | 14.63 eV | 30.50 eV |
| 51 | Ionization energy of Antimony (Sb) | 8.60 eV | 16.53 eV | 25.3 eV |
| 52 | Ionization energy of Tellurium (Te) | 9 eV | 18.6 eV | 27.96 eV |
| 53 | Ionization energy of Iodine (I) | 10.45 eV | 19.13 eV | 33 eV |
| 54 | Ionization energy of Xenon (Xe) | 12.12 eV | 21.20 eV | 32.12 eV |
| 55 | Ionization energy of Caesium (Cs) | 3.89 eV | 23.15 eV | – |
| 56 | Ionization energy of Barium (Ba) | 5.21 eV | 10 eV | – |
| 57 | Ionization energy of Lanthanum | 5.57 eV | 11.06 eV | 19.17 eV |
| 58 | Ionization energy of Cerium (Ce) | 5.53 eV | 10.85 eV | 20.19 eV |
| 59 | Ionization energy of Praseodymium (Pr) | 5.47 eV | 10.55 eV | 21.62 eV |
| 60 | Ionization energy of Neodymium (Nd) | 5.52 eV | 10.73 eV | 22.21 eV |
| 61 | Ionization energy of Promethium (Pm) | 5.58 eV | 10.9 eV | 22.3 eV |
| 62 | Ionization energy of Samarium (Sm) | 5.64 eV | 11.07 eV | 23.4 eV |
| 63 | Ionization energy of Europium (Eu) | 5.67 eV | 11.24 eV | 24.92 eV |
| 64 | Ionization energy of Gadolinium (Gd) | 6.15 eV | 12.09 eV | 20.63 eV |
| 65 | Ionization energy of Terbium (Tb) | 5.86 eV | 11.52 eV | 21.91 eV |
| 66 | Ionization energy of Dysprosium (Dy) | 5.93 eV | 11.67 eV | 22.8 eV |
| 67 | Ionization energy of Holmium (Ho) | 6.02 eV | 11.80 eV | 22.84 eV |
| 68 | Ionization energy of Erbium (Er) | 6.10 eV | 11.93 eV | 22.74 eV |
| 69 | Ionization energy of Thulium (Tm) | 6.18 eV | 12.05 eV | 23.63 eV |
| 70 | Ionization energy of Ytterbium (Yb) | 6.25 eV | 12.17 eV | 25.05 eV |
| 71 | Ionization energy of Lutetium (Lu) | 5.42 eV | 13.9 eV | 20.95 eV |
| 72 | Ionization energy of Hafnium (Hf) | 6.82 eV | 14.9 eV | 23.3 eV |
| 73 | Ionization energy of Tantalum (Ta) | 7.54 eV | – | – |
| 74 | Ionization energy of Tungsten (W) | 7.86 eV | – | – |
| 75 | Ionization energy of Rhenium (Re) | 7.83 eV | – | – |
| 76 | Ionization energy of Osmium (Os) | 8.43 eV | – | – |
| 77 | Ionization energy of Iridium (Ir) | 8.96 eV | – | – |
| 78 | Ionization energy of Platinum (Pt) | 8.95 eV | 18.56 eV | 28 eV |
| 79 | Ionization energy of Gold (Au) | 9.22 eV | 20.5 eV | 30 eV |
| 80 | Ionization energy of Mercury (Hg) | 10.43 v | 18.75 eV | 34.2 eV |
| 81 | Ionization energy of Thallium (Tl) | 6.10 eV | 20.42 eV | 29.83 eV |
| 82 | Ionization energy of Lead (Pb) | 7.41 eV | 15.03 eV | 1.93 eV |
| 83 | Ionization energy of Bismuth (Bi) | 7.28 eV | 16.69 eV | 25.56 eV |
| 84 | Ionization energy of Polonium (Po) | 8.41 eV | – | – |
| 85 | Ionization energy of Astatine (At) | 9.31 eV | – | – |
| 86 | Ionization energy of Radon (Rn) | 10.74 eV | – | – |
| 87 | Ionization energy of Francium (Fr) | 4.07 eV | – | – |
| 88 | Ionization energy of Radium (Ra) | 5.27 eV | 10.14 eV | – |
| 89 | Ionization energy of Actinium (Ac) | 5.17 eV | 12.1 eV | – |
| 90 | Ionization energy of Thorium (Th) | 6.30 eV | 11.5 eV | 20 eV |
| 91 | Ionization energy of Protactinium (Pa) | 5.89 eV | – | – |
| 92 | Ionization energy of Uranium (U) | 6.19 eV | – | – |
| 93 | Ionization energy of Neptunium (Np) | 6.26 eV | – | – |
| 94 | Ionization energy of Plutonium (Pu) | 6.02 eV | – | – |
| 95 | Ionization energy of Americium (Am) | 5.97 eV | – | – |
| 96 | Ionization energy of Curium (Cm) | 5.99 eV | – | – |
| 97 | Ionization energy of Berkelium (Bk) | 6.19 eV | – | – |
| 98 | Ionization energy of Californium (Cf) | 6.28 eV | – | – |
| 99 | Ionization energy of Einsteinium (Es) | 6.42 eV | – | – |
| 100 | Ionization energy of Fermium (Fm) | 6.50 v | – | – |
| 101 | Ionization energy of Mendelevium (Md) | 6.58 eV | – | – |
| 102 | Ionization energy of Nobelium (No) | 6.65 eV | – | – |
| 103 | Ionization energy of Lawrencium (Lr) | 4.9 eV | – | – |
| 104 | Ionization energy of Rutherfordium (Rf) | 6 eV | – | – |
| 105 | Ionization energy of Dubnium (Db) | – | – | – |
| 106 | Ionization energy of Seaborgium (Sg) | – | – | – |
| 107 | Ionization energy of Bohrium (Bh) | – | – | – |
| 108 | Ionization energy of Hassium (Hs) | – | – | – |
| 109 | Ionization energy of Meitnerium (Mt) | – | – | – |
| 110 | Ionization energy of Darmstadtium (Ds) | – | – | – |
| 111 | Ionization energy of Roentgenium (Rg) | – | – | – |
| 112 | Ionization energy of Copernicium (Cn) | – | – | – |
| 113 | Ionization energy of Nihonium (Nh) | – | – | – |
| 114 | Ionization energy of Flerovium (Fl) | – | – | – |
| 115 | Ionization energy of Moscovium (Mc) | – | – | – |
| 116 | Ionization energy of Livermorium (Lv) | – | – | – |
| 117 | Ionization energy of Tennessine (Ts) | – | – | – |
| 118 | Ionization energy of Oganesson (Og) | – | – | – |
External resources:
- Ionization energies of the elements (data page) – Wikipedia. (2020, September 1). Ionization Energies of the Elements (Data Page) – Wikipedia. https://en.wikipedia.org/wiki/Ionization_energies_of_the_elements_(data_page)
- NIST atomic spectra database. (n.d.). https://physics.nist.gov/PhysRefData/ASD/ionEnergy.html
- Ionization Energy and Electron Affinity. (n.d.). Ionization Energy and Electron Affinity. https://chemed.chem.purdue.edu/genchem/topicreview/bp/ch7/ie_ea.html
- Ionization energies. (n.d.). http://hyperphysics.phy-astr.gsu.edu/hbase/Chemical/ionize.html
- Ionization Energy and Electronegativity. (n.d.). Ionization Energy and Electronegativity. http://butane.chem.uiuc.edu/pshapley/GenChem2/Intro/3.html
- Ionization Energy Search. (n.d.). Ionization Energy Search. https://webbook.nist.gov/chemistry/ie-ser/
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
