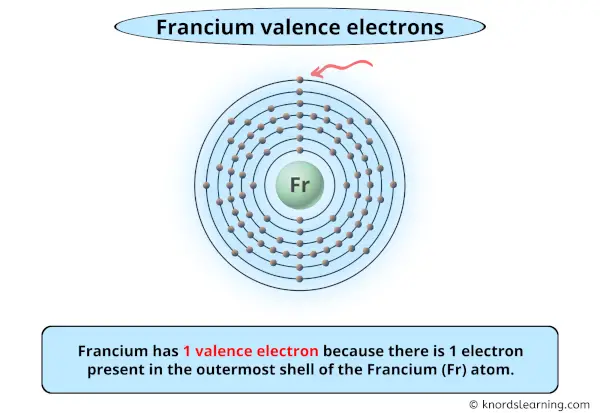
So you have seen the above image by now, right?
Awesome! You can see that francium has 1 valence electron.
But how can you say that Francium has 1 valence electron
+
How can you find this valence electron?
Let’s discuss this in short.
Francium has 1 valence electron because there is 1 electron present in the outermost shell of the Francium (Fr) atom.
Now let’s see how you can easily find the valence electrons of Francium atom (Fr).
If you don’t want to read the texts, then you can also watch this video.
How to find the Valence Electrons? (2 Methods)
In order to find the valence electrons of Francium atom (Fr), you can use two methods.
Method 1: From the Periodic Table
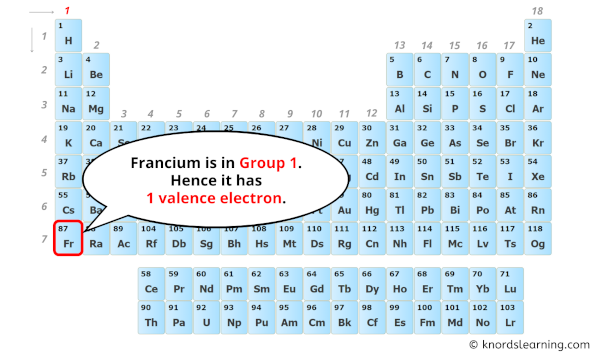
To find out the valence electrons of Francium, you have to see the position of francium in the periodic table.
More specifically, you have to see the group wise position of Francium element in the periodic table.
From the above image, you can see that Francium (Fr) is present in the group 1 of periodic table.
(Note: Group 1 is also called group 1A).
So, as the francium element is present in group 1, it has 1 valence electron.
In this way, by knowing the position of francium element in periodic table, you can easily find its valence electrons.
Now let’s see another method for finding the number of valence electrons in francium.
Method 2: From the Electron Configuration
If you want to find the valence electrons of francium from its electron configuration, then you should know its electron configuration first.
Now there are many methods to write the electron configurations, but here I will show you the easiest method, i.e by using Aufbau principle.
Aufbau principle: The Aufbau principle simply states that the orbitals with the lower energy are filled first and then the orbitals with higher energy levels are filled.
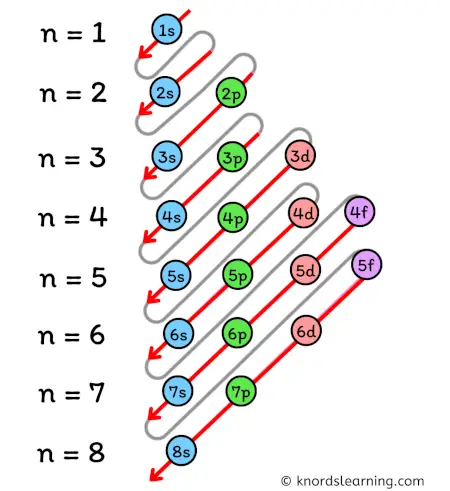
According to the Aufbau principle, the orbitals are filled in the following order:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on.
Also the maximum number of electrons that can be accommodated in s, p, d & f orbitals are mentioned in the below table.
| Orbitals | Maximum capacity of electrons [1] |
| s | 2 |
| p | 6 |
| d | 10 |
| f | 14 |
Now let’s try to find the electron configuration of Francium by using the Aufbau principle.
Electron Configuration of Francium:
Follow the steps mentioned below to get the electron configuration of Francium.
- To write the electron configuration of francium, we should first know the total number of electrons present in a francium atom.
- The francium atom has a total of 87 electrons because its atomic number is 87 and it is a neutral atom. [2]
- Now we have to fill these 87 electrons in the atomic orbitals according to the Aufbau principle.
- According to the Aufbau principle, the electrons will be filled first in 1s orbital, then in 2s orbital, then in 2p orbital, and so on…
- So from the Aufbau principle, we can get the electron configuration of the francium atom as 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f14 5d10 6s2 6p6 7s1. [3]
Now in this electron configuration of francium, we have to see the total number of electrons present in the highest energy level.
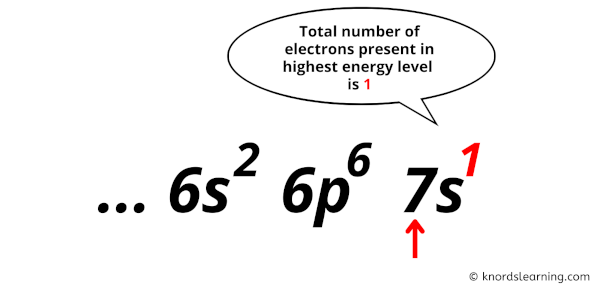
You can see in the electron configuration of francium (1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f14 5d10 6s2 6p6 7s1) that the highest energy level is 7. And the total number of electrons present in this energy level is 1.
So by knowing the electron configuration, we have found that the Francium has 1 valence electron.
I hope you have understood the methods of finding the valence electrons in francium.
See more related topics for your practice;
Radium Valence Electrons
Boron Valence Electrons
Carbon Valence Electrons
Nitrogen Valence Electrons
Oxygen Valence Electrons
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
