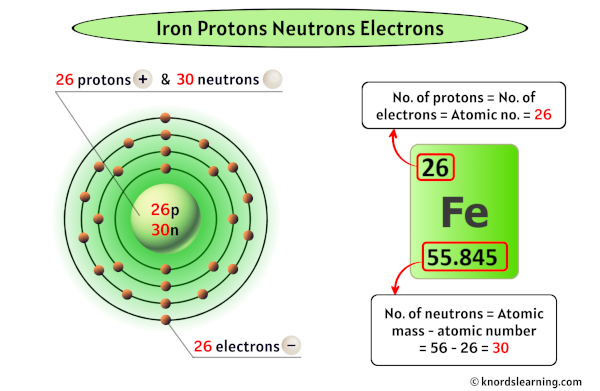
Iron has 26 protons, 30 neutrons and 26 electrons.
But how will you find the number of protons, neutrons and electrons in Iron (Fe)?
Well, it is very easy to find the protons, neutrons and electrons of iron atom.
Here I have given a very simple method for finding the protons, neutrons and electrons of iron atom.
Let’s dive right into it!
Finding the Protons, Neutrons and Electrons in Iron
How to find protons?
- The number of protons can be found by knowing the atomic number of that atom. [1]
How to find neutrons?
- The number of neutrons can be found by subtracting the atomic number from its atomic mass.
How to find electrons?
- For a neutral atom, the number of electrons can be found by knowing the atomic number of that atom.
Let’s calculate the number of protons, neutrons and electrons in iron.
If you don’t want to read, then you can also watch this video.
#1 Number of Protons in Iron
If you have a periodic table with you, then most of the answers are in front of you.

You can see the elements, their atomic number and their atomic mass from the periodic table.
Now here our element is Iron (Fe).
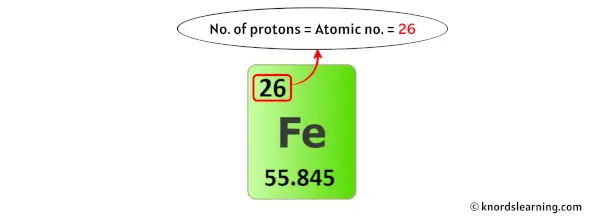
So from the above periodic table, you can see that the atomic number of iron is 26.
As the atomic number of iron is 26, it has a total of 26 protons in its nucleus.
Thus, the number of protons in Iron is 26.
#2 Number of Neutrons in Iron
In order to find the number of neutrons of iron atom, you should know its atomic mass first.
The number of neutrons in iron can be obtained by subtracting the atomic number from its atomic mass.

The atomic mass of iron is 55.845 u (which you can round it to 56). [2]
So from this atomic mass (i.e 56), you have to subtract its atomic number (i.e 26).
So you will get 56 – 26 = 30.
Thus, the number of neutrons in Iron is 30.
#3 Number of Electrons in Iron
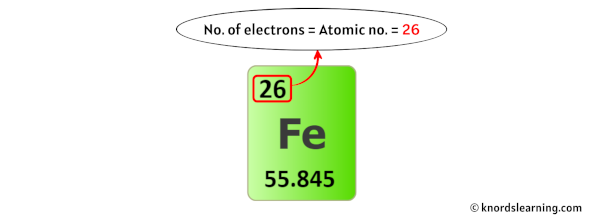
For a neutral atom, the number of electrons and the number of protons are the same.
Here, the iron atom is a neutral atom.
So the number of electrons in iron is equal to its number of protons (which is also equal to its atomic number).
In the beginning, we have found that the number of protons in iron is 26.
Thus, the number of electrons in Iron is 26.
Summary
Number of Protons in Iron
- The number of protons can be found by knowing the atomic number of that atom.
- Number of Protons in Iron = Atomic number of Iron = 26
Number of Neutrons in Iron
- The number of neutrons can be found by subtracting the atomic number from its atomic mass.
- Number of Neutrons in Iron = Atomic mass of Iron – Atomic number of Iron = 56 – 26 = 30
Number of Electrons in Iron
- For a neutral atom, the number of electrons can be found by knowing the atomic number of that atom.
- Number of Electrons in Iron = Atomic number of Iron = 26
I hope you have understood the simple method for finding the protons, neutrons and electrons of iron atom.
Check out related topics for more practice;
Cobalt protons neutrons electrons
Nickel protons neutrons electrons
Copper protons neutrons electrons
Zinc protons neutrons electrons
Zirconium protons neutrons electrons
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
