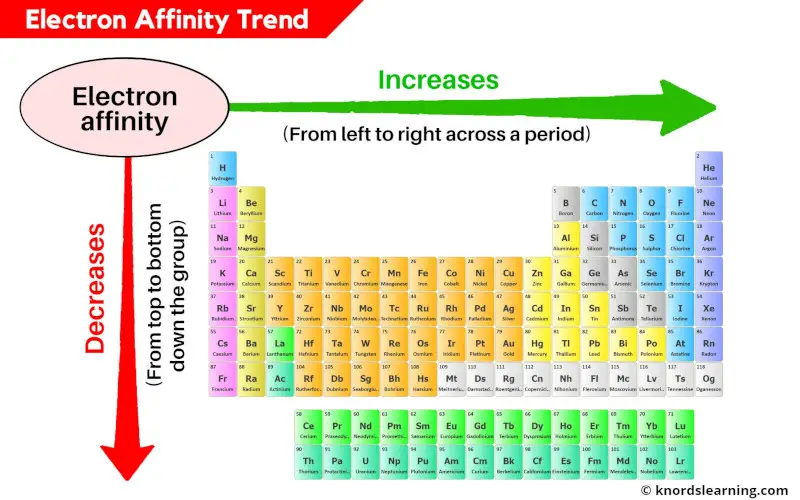
Electron Affinity Trend:
Across the period (from left to right): Increases
Down the group (from top to bottom): Decreases
The electron affinity of elements increases as we move across a period (from left to right) and it decreases as we move down in a group (from top to bottom).
Well this was just an introduction about the electron affinity trends in periodic table.
But you need to understand why the electron affinity trends of elements increase across a period and why it decreases down the group.
So let’s dive right into it!
Explanation about Electron Affinity Trends
Before understanding the electron affinity trends, you should know what electron affinity is?
Definition: Electron affinity is defined as the amount of energy released when an electron is added in the outermost shell of an isolated gaseous atom.
In simple words, electron affinity is the tendency of an atom to accept the electron.
Now let’s understand the electron affinity trend in periodic table.
There are many factors that affect the electron affinity trend. But I’ll give you the simple basic explanation that is enough to understand the electron affinity trend.
You know that the nucleus of an atom contains protons which are positively charged particles. So these positively charged particles have a tendency to attract the negatively charged electrons which are around the nucleus.
Now you might be knowing the trends of atomic radius on the periodic table.
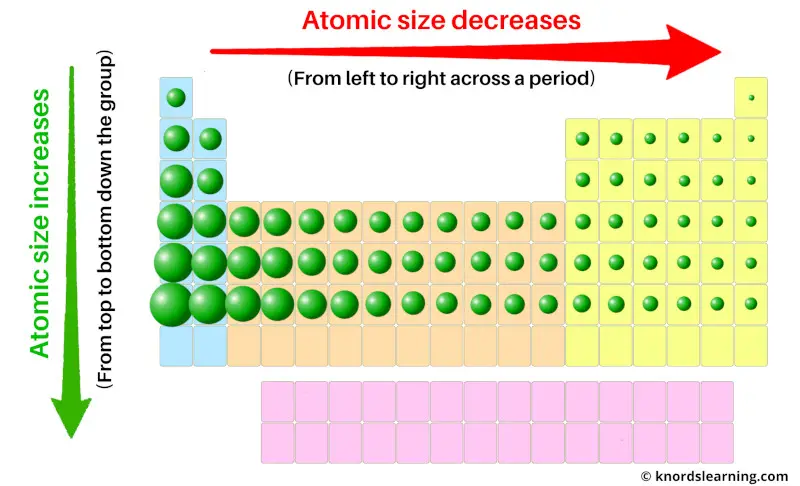
If we move across the period (from left to right), then the atomic size decreases. And if we move down the group (from top to bottom), then the atomic size increases.
Now, what does a smaller atomic size indicate? And what does a larger atomic size indicate?
The smaller atomic size indicates that it has more tendency to accept the free electron.
So from this we can say that;
Less the atomic size, more will be the electron affinity (tendency to accept the electron).
In simple words, electron affinity is inversely proportional to the atomic size.
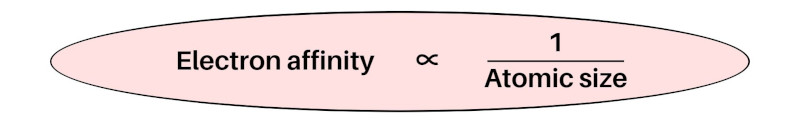
As we move across the period (from left to right), the atomic size decreases, so the electron affinity increases.
And similarly as we move down the group (from top to bottom), the atomic size increases, so the electron affinity decreases.
I hope you have understood the reason behind the electron affinity trend on the periodic table.
Checkout other trends in periodic table:
- Atomic radius trend
- Electronegativity trend
- Ionization energy trend
- Metallic character trend
- Non metallic character trend
External resources:
- McCord, P. (n.d.). Periodic Table Trends. Periodic Table Trends. https://mccord.cm.utexas.edu/chembook/page.php?chnum=3§=10
- Boudreaux, K. A. (n.d.). The Parts of the Periodic Table. The Parts of the Periodic Table. https://www.angelo.edu/faculty/kboudrea/periodic/trends_summary.htm
- Periodic Table: Trends. (n.d.). Periodic Table: Trends. https://www.rsc.org/periodic-table/trends
- Periodic trends – Wikipedia. (2022, July 2). Periodic Trends – Wikipedia. https://en.wikipedia.org/wiki/Periodic_trends
- Periodic Trends. (2013, October 2). Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Supplemental_Modules_and_Websites_(Inorganic_Chemistry)/Descriptive_Chemistry/Periodic_Trends_of_Elemental_Properties/Periodic_Trends
- University of Alabama in Huntsville. https://www.uah.edu/images/administrative/student-success-center/resources/handouts/handouts_2019/periodic_trends.pdf
- Americal Chemical Society. https://www.acs.org/content/dam/acsorg/education/students/highschool/chemistryclubs/infographics/mastering-periodic-trends-infographic.pdf
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
