
Periodic table with metals, nonmetals and metalloids shows that the metals are on the left side, nonmetals are on the right side and metalloids are forming a zigzag line between them.
Well in this article, you will also get to know the total number of metals, nonmetals and metalloids present on the periodic table.
So let’s dive right into it!
Periodic Table Metals
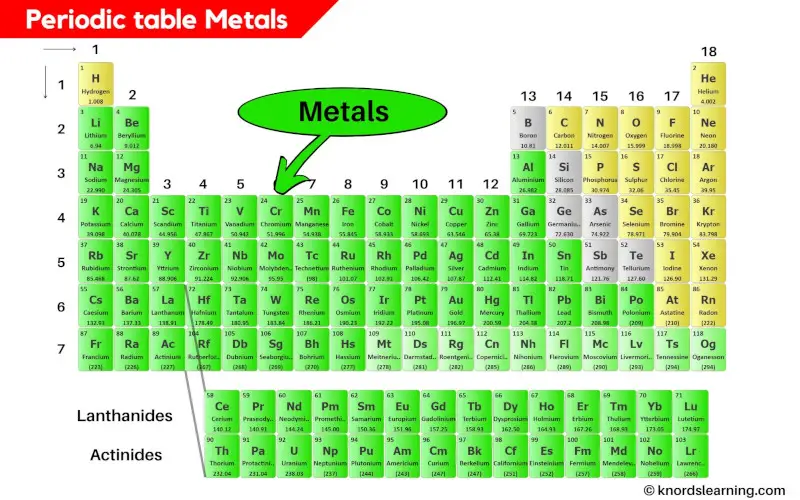
Metals are the elements on the periodic table that are usually hard, shiny and they easily conduct heat and electricity through it.
They are ductile as well as malleable, which means they can be drawn into thin sheets and wires.
There are many different types of metals on the periodic table like; Alkali metals, Alkaline earth metals, Transition metals, Inner transition metals, Post transition metals, Rare earth metals, Heavy metals, etc. You can read about all these different metals from this article.
How many Metals are on the periodic table?
There are around 94 metals on the periodic table.
This means that around 78% of the elements on the periodic table are metals.
But this number is not exact.
Why?
Because the boundaries between metals, nonmetals and metalloids fluctuate slightly due to lack of universally accepted definitions.
In metallurgy, the researchers define the metals on the basis of density. In chemistry, the chemists define the metals on the basis of chemical properties. While in physics, the researchers may define the metals on the basis of physical properties.
So there is no universally accepted definition for metals.
Also, the elements 109 to 118 have a very short half-life and they are still under research.
So if we considered the first 108 elements, then there are 84 known metals on the periodic table.
Periodic Table Nonmetals
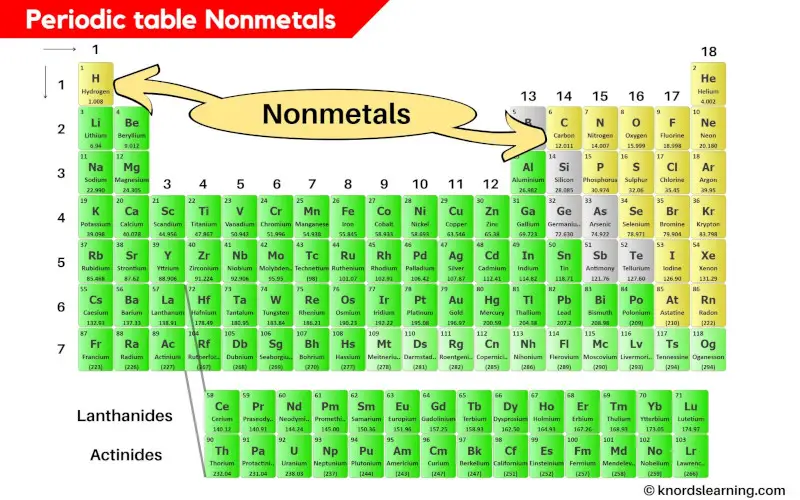
Nonmetals are the elements on the periodic table that are soft and dull.
They do not have a lustrous surface and they are bad conductors of heat and electricity.
Nonmetals are located on the right side of the periodic table (hydrogen is the only nonmetal that is on the left-top corner of the periodic table.)
During a chemical reaction, the nonmetals gain electrons to complete the octet.
How many Nonmetals are on the periodic table?
There are a total of 18 Nonmetals on the periodic table.
Element 85 (Astatine) shows some properties of nonmetals as well as some properties of metalloids.
But astatine is classified as a nonmetal on periodic table. (Read the reason why astatine is classified as a nonmetal).
Periodic Table Metalloids
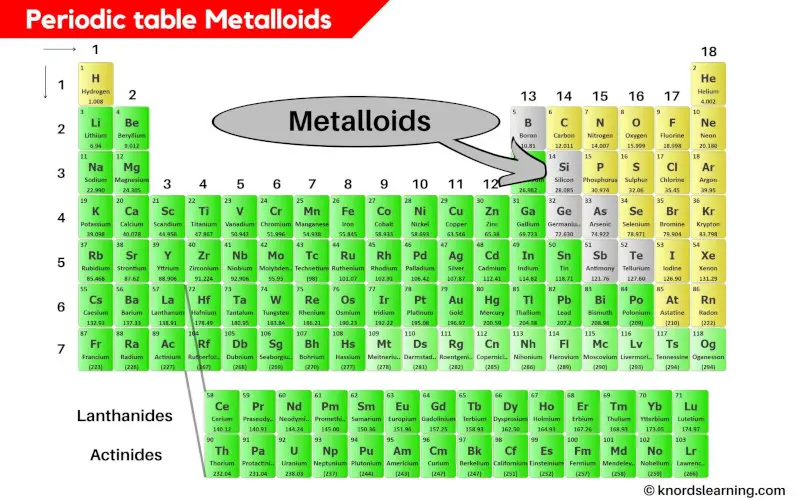
Metalloids are present between the metals and nonmetals on the periodic table.
The metalloids are the elements that have properties that are intermediate between those of metals and nonmetals.
As the metalloids have few properties of metals and few properties of nonmetals, they are also known as semimetals.
How many Metalloids are on the periodic table?
There are 6 commonly known metalloids on the periodic table.
But this number is not exact as there is no universally accepted definition for metalloids.
Astatine and polonium show few properties of metalloids but they are not classified as metalloids. Because astatine shows more properties of metals and polonium shows more properties of halogens.
External resources:
- Metals and Nonmetals. GSU. http://hyperphysics.phy-astr.gsu.edu/hbase/pertab/metal.html
- 6.7: Metalloids. (2016, June 27). Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introductory_Chemistry_(CK-12)/06%3A_The_Periodic_Table/6.07%3A_Metalloids
- Metal – Wikipedia. (2021, March 25). Metal – Wikipedia. https://en.wikipedia.org/wiki/Metal
- Boudreaux, K. A. (n.d.). The Parts of the Periodic Table. The Parts of the Periodic Table. https://www.angelo.edu/faculty/kboudrea/periodic/physical_metals.htm
- What is a Metal ? (n.d.). https://depts.washington.edu/matseed/mse_resources/Webpage/Metals/metals.htm
- The Chemistry of Nonmetals. (n.d.). The Chemistry of Nonmetals. https://chemed.chem.purdue.edu/genchem/topicreview/bp/ch10/non.php
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
