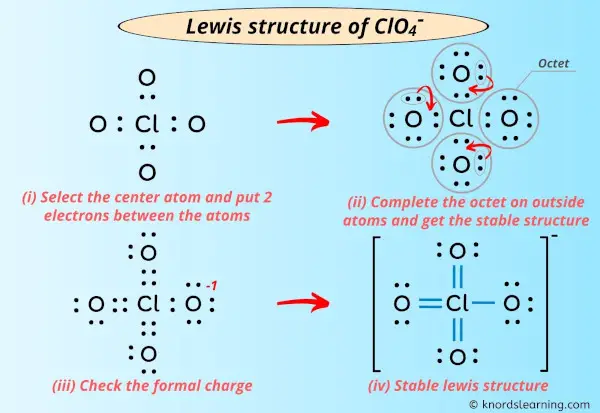
I’m super excited to teach you the lewis structure of ClO4- ion in just 5 simple steps.
Infact, I’ve also given the step-by-step images for drawing the lewis dot structure of ClO4- ion.
So, if you are ready to go with these 5 simple steps, then let’s dive right into it!
Lewis structure of ClO4- ion (Perchlorate ion) contains three double bonds and one single bond between the Chlorine (Cl) atom and Oxygen (O) atoms. The Chlorine atom (Cl) is at the center and it is surrounded by 4 Oxygen atoms (O).
Let’s draw and understand this lewis dot structure step by step.
(Note: Take a pen and paper with you and try to draw this lewis structure along with me. I am sure you will definitely learn how to draw lewis structure of ClO4– ion).
5 Steps to Draw the Lewis Structure of ClO4-
Step #1: Calculate the total number of valence electrons
Here, the given ion is ClO4– (perchlorate ion). In order to draw the lewis structure of ClO4– ion, first of all you have to find the total number of valence electrons present in the ClO4– ion.
(Valence electrons are the number of electrons present in the outermost shell of an atom).
So, let’s calculate this first.
Calculation of valence electrons in ClO4- ion
- For Chlorine:
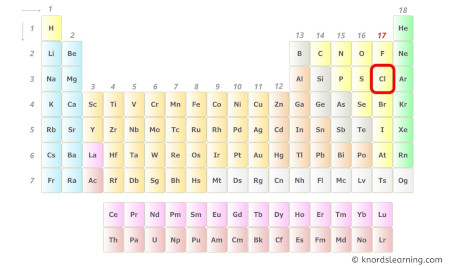
Chlorine is a group 17 element on the periodic table. [1]
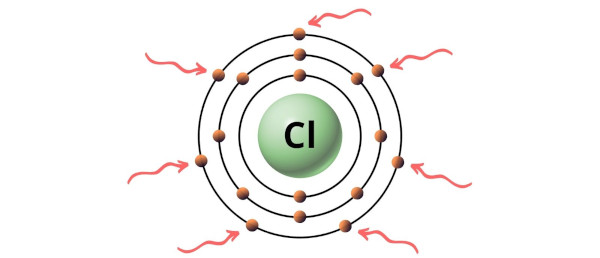
Hence, the valence electron present in chlorine is 7 (see below image).
- For Oxygen:
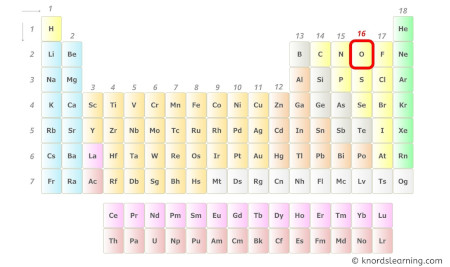
Oxygen is a group 16 element on the periodic table. [2]
Hence, the valence electron present in oxygen is 6 (see below image).
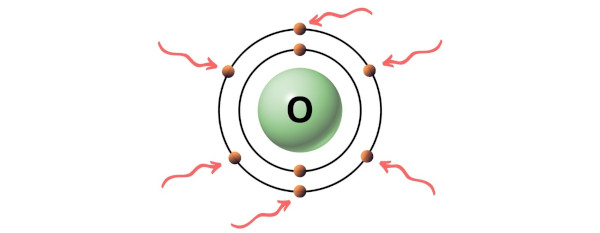
Hence in a ClO4- ion,
Valence electrons given by Chlorine (Cl) atom = 7
Valence electrons given by each Oxygen (O) atom = 6
Electron due to -1 charge, 1 more electron is added
So, total number of Valence electrons in ClO4– ion = 7 + 6(4) + 1 = 32
Step #2: Select the center atom
While selecting the center atom, always put the least electronegative atom at the center.
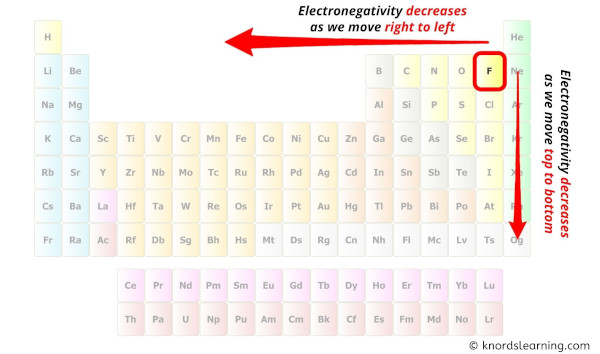
(Remember: Fluorine is the most electronegative element on the periodic table and the electronegativity decreases as we move right to left in the periodic table as well as top to bottom in the periodic table). [3]
Here in the ClO4 molecule, if we compare the chlorine atom (Cl) and oxygen atom (O), then chlorine is less electronegative than oxygen.
So, chlorine should be placed in the center and the remaining 4 oxygen atoms will surround it.
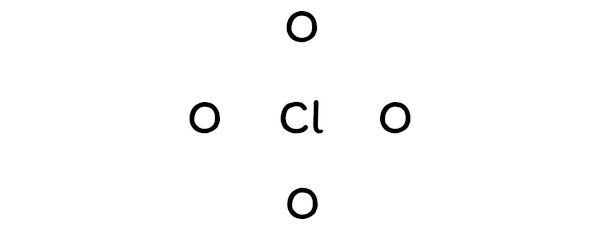
Step #3: Put two electrons between the atoms to represent a chemical bond
Now in the above sketch of ClO4, put the two electrons (i.e electron pair) between each chlorine atom and oxygen atom to represent a chemical bond between them.
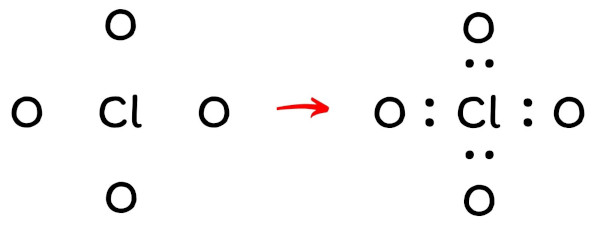
These pairs of electrons present between the Chlorine (Cl) and Oxygen (O) atoms form a chemical bond, which bonds the chlorine and oxygen atoms with each other in a ClO4– ion.
Step #4: Complete the octet (or duplet) on outside atoms
In the Lewis structure of ClO4– ion, the outer atoms are oxygen atoms.
So now, you have to complete the octet on these oxygen atoms (because oxygen requires 8 electrons to have a complete outer shell).
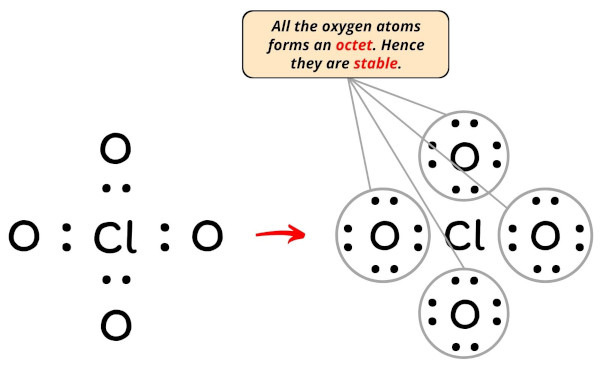
Now, you can see in the above image that all the oxygen atoms form an octet.
Also, all the 32 valence electrons of ClO4– ion (as calculated in step #1) are used in the above structure. So there are no remaining electron pairs.
Hence there is no change in the above sketch of ClO4– ion.
Let’s move to the next step.
Step #5: Check the formal charge
You can see from the above image that the central atom (i.e chlorine), is having 8 electrons. So it fulfills the octet rule.
But, in order to get the most stable lewis structure, we have to check the formal charge on ClO4– ion.
For that, you need to remember the formula of formal charge;
Formal charge = Valence electrons – Nonbonding electrons – (Bonding electrons)/2
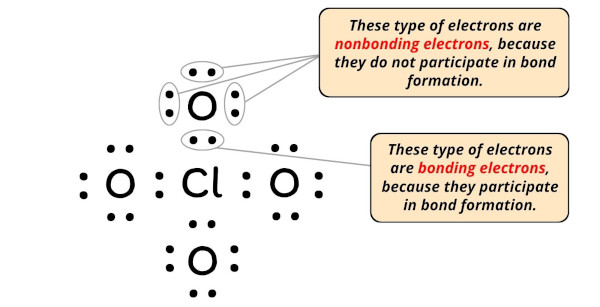
- For Chlorine:
Valence electrons = 7 (as it is in group 17)
Nonbonding electrons = 0
Bonding electrons = 8 - For Oxygen:
Valence electron = 6 (as it is in group 16)
Nonbonding electrons = 6
Bonding electrons = 2
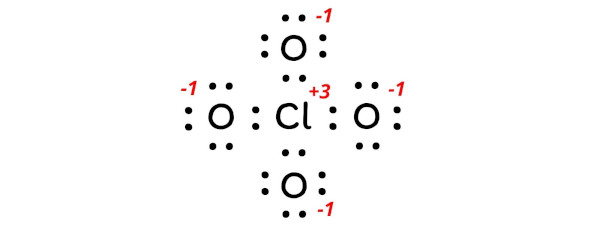
| Formal charge | = | Valence electrons | – | Nonbonding electrons | – | (Bonding electrons)/2 | ||
| Cl | = | 7 | – | 0 | – | 8/2 | = | +3 |
| O | = | 6 | – | 6 | – | 2/2 | = | -1 |
Let’s keep these charges on the atoms in the above lewis structure of ClO4– ion.
As we know that chlorine can hold more than 8 electrons, we can further reduce the charges as mentioned below.
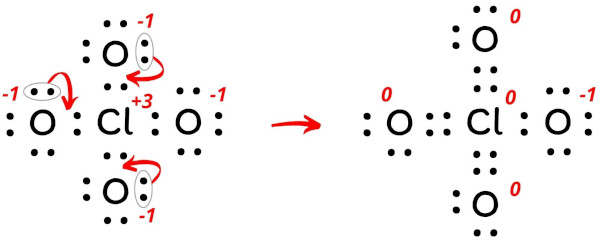
By doing this, the formal charges on the three oxygen atoms as well as the central chlorine atom becomes “zero”, and this gives us the most stable lewis structure of ClO4-.
There is one -ve charge left on the oxygen atom, which indicates the -1 formal charge on the ClO4– ion.
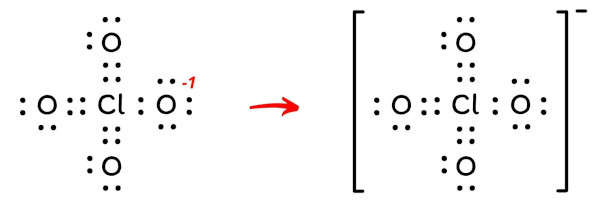
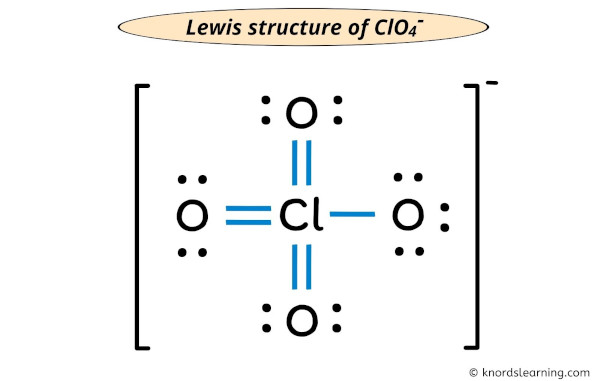
Each electron pair (:) in the lewis dot structure of ClO4– ion represents the single bond ( | ). So the above lewis dot structure of ClO4– ion can also be represented as shown below.
Related lewis structures for your practice:
Lewis structure of ClO-
Lewis structure of NOCl
Lewis structure of SeO2
Lewis structure of OCl2
Lewis structure of CH3COOH
Article by;
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
