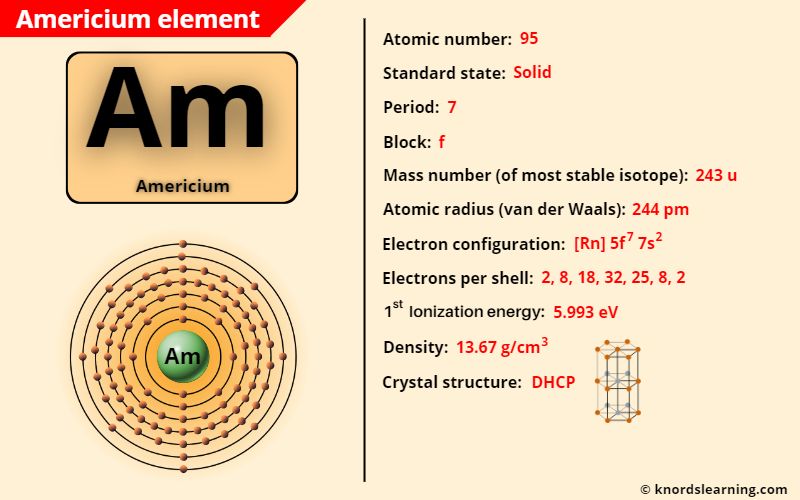
Americium element (Am) is in period 7 of a periodic table. Americium is in the f-block and it is classified as an actinide on the periodic table.
There is a lot more information related to americium which is mentioned in the Information Table given below.
So let’s dive right into it!
Table of contents
- Americium element (Information Table)
- Americium element in Periodic table
- Facts about Americium
- Properties of Americium
- Uses of Americium
Americium Element (Information Table)
The important data related to americium element is given in the table below.
| Appearance of americium | Silvery white metallic appearance |
| Atomic number of americium | 95 |
| Symbol of americium | Am |
| Atomic mass of americium (most stable isotope) | 243 u |
| Protons, Neutrons & Electrons in americium | Protons: 95, Neutrons: 148, Electrons: 95 |
| State of americium (at STP) | Solid |
| Period number of americium in periodic table | 7 |
| Block of americium in periodic table | f-block |
| Category of americium | Inner transition metals |
| Bohr model or Electrons per shell or Electrons arrangement in americium | 2, 8, 18, 32, 25, 8, 2 |
| Electron configuration of americium | [Rn] 5f7 7s2 |
| Orbital diagram of americium | 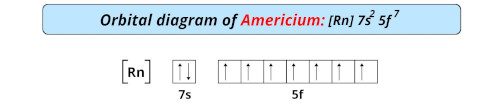 |
| Atomic radius of americium (van der Waals radius) | 244 picometers |
| Density of americium | 13.67 g/cm3 |
| 1st ionization energy of americium | 5.993 eV |
| Main isotope of americium | 241Am and 243Am |
| Melting point of americium | 1449 K or 1176 °C or 2149 °F |
| Boiling point of americium (calculated values) | 2880 K or 2607 °C or 4725 °F |
| Crystal structure of americium | Double hexagonal close packed |
| Discovery of americium | By Glenn T. Seaborg, Albert Ghiorso, Leon O. Morgan, Ralph A. James (in 1944) |
Also see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table).
Americium element in Periodic table
The Americium element (Am) has the atomic number 95 and is located in period 7. Americium is a metal and it is classified as an actinide group element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Click on above elements in the periodic table to see their information.
Facts about americium
Here are a few interesting facts about the americium element.
- Glenn T. Seaborg, Albert Ghiorso, Leon O. Morgan and Ralph A. James discovered the americium element in the year 1944.
- Americium was given its name from “America”.
- There are around 19 isotopes of americium and all these isotopes are radioactive in nature.
- 243Am is the isotope of americium which is most stable and has a half life of 7370 years.
- Americium is naturally available from uranium ores, but this naturally available uranium is very rare in quantity. Most of the americium is produced artificially in the lab.
Properties of americium
Here is a list of some physical properties and chemical properties of americium.
Physical properties of americium
- Americium has a silvery white appearance.
- Americium has double hexagonal close packed crystal structure.
- The melting point of americium is 1449 K and its boiling point is 2880 K (these are calculated values).
Chemical properties of americium
- Americium is a toxic and radioactive metal.
- Pure americium reacts with the atmospheric oxygen and starts tarnishing.
- The most common oxidation state of americium is +3. Apart from this, it also has other oxidation states ranging from +2 to +7.
Uses of americium
Here are some uses of the americium element.
- 241Am is the isotope of americium (which is present in the compound americium dioxide) is used in household smoke detectors.
- 241Am is also used in some industrial applications as well as some medical applications.
- Americium can also be used to produce other transuranic elements.
External resources:
- Americium – Wikipedia. (2023, February 11). Americium – Wikipedia. https://en.wikipedia.org/wiki/Americium
- Americium – Element information, properties and uses | Periodic Table. (n.d.). Americium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/95/americium
- Bondi, A. (1964, March). van der Waals Volumes and Radii. The Journal of Physical Chemistry, 68(3), 441–451. https://doi.org/10.1021/j100785a001
- Holden, et al. (2018, December 1). IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Education Community (IUPAC Technical Report). Pure and Applied Chemistry, 90(12), 1833–2092. https://doi.org/10.1515/pac-2015-0703
- Zhang, et al. (2011, January 11). Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. Journal of Chemical & Engineering Data, 56(2), 328–337. https://doi.org/10.1021/je1011086
- P. (n.d.). Americium | Am (Element) – PubChem. Americium | Am (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Americium
- It’s Elemental – The Element Americium. (n.d.). It’s Elemental – the Element Americium. https://education.jlab.org/itselemental/ele095.html
- Radionuclide Basics: Americium-241 | US EPA. (2015, April 14). US EPA. https://www.epa.gov/radiation/radionuclide-basics-americium-241
- Americium | CCDC. (n.d.). Americium | CCDC. https://www.ccdc.cam.ac.uk/elements/americium/
- Prohaska, T., et al. (2022, May 1). Standard atomic weights of the elements 2021 (IUPAC Technical Report). Pure and Applied Chemistry, 94(5), 573–600. https://doi.org/10.1515/pac-2019-0603
- Haynes, W. M. (Ed.). (2014, June 4). CRC Handbook of Chemistry and Physics. https://doi.org/10.1201/b17118
- Kaye, G W.C., & Laby, T H. Tables of physical and chemical constants. 15th Edition. United States.
- Sansonetti, J. E., & Martin, W. C. (2005, December). Handbook of Basic Atomic Spectroscopic Data. Journal of Physical and Chemical Reference Data, 34(4), 1559–2259. https://doi.org/10.1063/1.1800011
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
