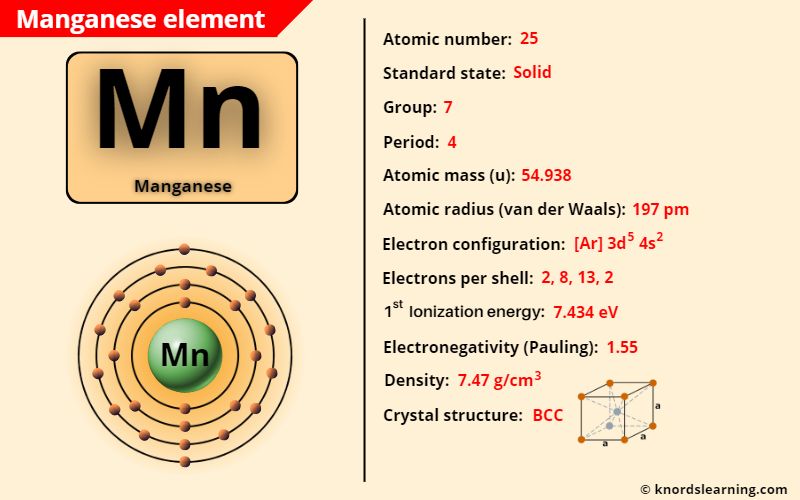
Manganese element (Mn) is in group 7 and period 4 of a periodic table. Manganese is in the d-block and it is classified as a transition element on the periodic table.
There is a lot more information related to manganese which is mentioned in the Information Table given below.
So let’s dive right into it!
Table of contents
- Manganese element (Information Table)
- Manganese element in Periodic table
- Facts about Manganese
- Properties of Manganese
- Uses of Manganese
Manganese Element (Information Table)
The important data related to manganese element is given in the table below.
| Appearance of manganese | Silvery white appearance |
| Atomic number of manganese | 25 |
| Symbol of manganese | Mn |
| Atomic mass of manganese | 54.938 u |
| Protons, Neutrons & Electrons in manganese | Protons: 25, Neutrons: 30, Electrons: 25 |
| State of manganese (at STP) | Solid |
| Group number of manganese in periodic table | 7 |
| Period number of manganese in periodic table | 4 |
| Block of manganese in periodic table | d-block |
| Category of manganese | Transition metals |
| Bohr model or Electrons per shell or Electrons arrangement in manganese | 2, 8, 13, 2 |
| Electron configuration of manganese | [Ar] 3d5 4s2 |
| Orbital diagram of manganese | 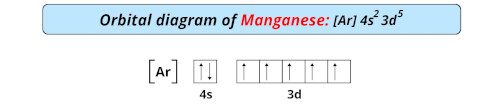 |
| Electronegativity of manganese (on pauling scale) | 1.55 |
| Atomic radius of manganese (van der Waals radius) | 197 picometers |
| Density of manganese | 7.47 g/cm3 |
| 1st ionization energy of manganese | 7.434 eV |
| Main isotope of manganese | 55Mn |
| Melting point of manganese | 1519 K or 1246 °C or 2275 °F |
| Boiling point of manganese | 2334 K or 2061 °C or 3742 °F |
| Crystal structure of manganese | Body Centered Cubic (BCC) |
| Discovery of manganese | By Carl Wilhelm Scheele in 1774 |
Also see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table).
Manganese element in Periodic table
The Manganese element (Mn) has the atomic number 25 and is located in group 7 and period 4. Manganese is a metal and it is classified as a transition element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Click on above elements in the periodic table to see their information.
Facts about manganese
Here are a few interesting facts about manganese element.
- Manganese is the 12th most abundant element found from the earth’s crust.
- Around 75% of the total manganese resources comes from South Africa, which makes it a leading producer of manganese in the entire world.
- Most of the manganese (around 85% to 90%) is used in manufacturing of steels.
- Manganese is brittle than iron, but it has more hardness than iron.
- The manganese occupies around 0.1% (by weight) of the earth’s crust.
- Manganese is also required by the human body to perform metabolic activities.
Properties of manganese
Here is a list of some physical properties and chemical properties of manganese.
Physical properties of manganese
- Manganese has a solid metal having a silvery white appearance.
- Manganese has a shiny surface when it is polished, but it gets tarnished easily when it is exposed to air.
- Manganese has a density of 7.47 g/cm3 and its atomic mass is 54.938 u.
- Manganese is not malleable because it is strong but brittle in nature.
- The melting point and boiling point of manganese is 1519 K and 2334 K respectively.
Chemical properties of manganese
- Manganese is a reactive metal and because of this reason it is not found in free state from the earth’s crust. It is always found as a compound with other elements.
- There are many isotopes of manganese. Out of these isotopes, the most abundant isotope is 55Mn.
- When pure manganese comes in contact with water, it easily gets corroded.
- The electronic configuration of manganese is [Ar] 3d5 4s2 which has incomplete d-orbitals.
Uses of manganese
Here are some uses of the manganese element.
- Manganese is widely used in steel manufacturing industries as well as it is also used in making glass and other materials.
- Manganese is used in aluminum alloys for manufacturing the cans for storage of beverages.
- Manganese is also present in the human body and our body requires manganese for proper metabolic activities. The human body gets the manganese from foods like eggs, spinach, rice, nuts, etc.
External resources:
- Manganese. (n.d.). Manganese. https://webbook.nist.gov/cgi/inchi/InChI%3D1S/Mn
- Manganese – Element information, properties and uses | Periodic Table. (n.d.). Manganese – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/25/manganese
- Manganese – Wikipedia. (2019, September 7). Manganese – Wikipedia. https://en.wikipedia.org/wiki/Manganese
- It’s Elemental – The Element Manganese. (n.d.). It’s Elemental – the Element Manganese. https://education.jlab.org/itselemental/ele025.html
- Atomic Weight of Manganese | Commission on Isotopic Abundances and Atomic Weights. (n.d.). Atomic Weight of Manganese | Commission on Isotopic Abundances and Atomic Weights. https://ciaaw.org/manganese.htm
- Atomic Data for Manganese (Mn). (n.d.). Atomic Data for Manganese (Mn). https://physics.nist.gov/PhysRefData/Handbook/Tables/manganesetable1.htm
- Manganese Statistics and Information | U.S. Geological Survey. (n.d.). Manganese Statistics and Information | U.S. Geological Survey. https://www.usgs.gov/centers/national-minerals-information-center/manganese-statistics-and-information
- Haynes, W. M. (Ed.). (2014, June 4). CRC Handbook of Chemistry and Physics. https://doi.org/10.1201/b17118
- Kaye, G W.C., & Laby, T H. Tables of physical and chemical constants. 15th Edition. United States.
- Sansonetti, J. E., & Martin, W. C. (2005, December). Handbook of Basic Atomic Spectroscopic Data. Journal of Physical and Chemical Reference Data, 34(4), 1559–2259. https://doi.org/10.1063/1.1800011
- Bondi, A. (1964, March). van der Waals Volumes and Radii. The Journal of Physical Chemistry, 68(3), 441–451. https://doi.org/10.1021/j100785a001
- Holden, et al. (2018, December 1). IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Education Community (IUPAC Technical Report). Pure and Applied Chemistry, 90(12), 1833–2092. https://doi.org/10.1515/pac-2015-0703
- Emsley, J. (2011). Nature’s Building Blocks: An A-Z Guide to the Elements. United Kingdom: OUP Oxford.
- Allred, A. (1961, June). Electronegativity values from thermochemical data. Journal of Inorganic and Nuclear Chemistry, 17(3–4), 215–221. https://doi.org/10.1016/0022-1902(61)80142-5
- James A. M. & Lord M. P. (1992). Macmillan’s chemical and physical data. Macmillan.
- Zhang, Y., Evans, J. R. G., & Yang, S. (2011, January 11). Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. Journal of Chemical & Engineering Data, 56(2), 328–337. https://doi.org/10.1021/je1011086
- Possolo, A., van der Veen, A. M. H., Meija, J., & Hibbert, D. B. (2018, January 4). Interpreting and propagating the uncertainty of the standard atomic weights (IUPAC Technical Report). Pure and Applied Chemistry, 90(2), 395–424. https://doi.org/10.1515/pac-2016-0402
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
