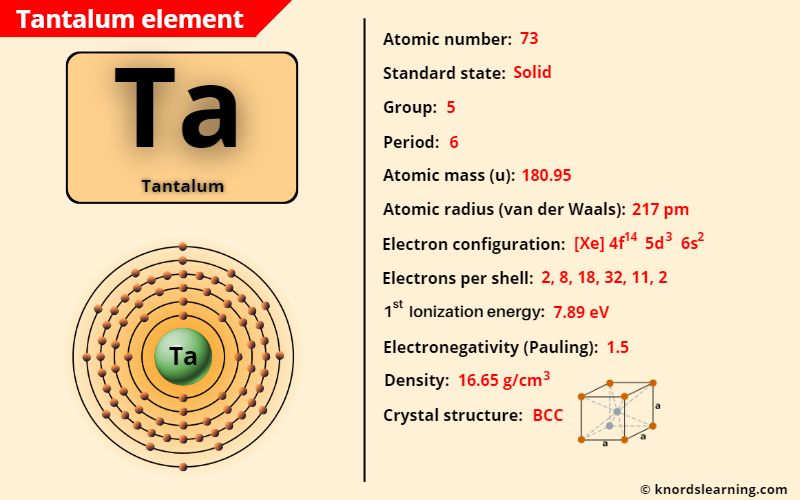
Tantalum element (Ta) is in group 5 and period 6 of a periodic table. Tantalum is in the d-block and it is classified as a transition element on the periodic table.
There is a lot more information related to tantalum which is mentioned in the Information Table given below.
So let’s dive right into it!
Table of contents
- Tantalum element (Information Table)
- Tantalum element in Periodic table
- Facts about Tantalum
- Properties of Tantalum
- Uses of Tantalum
Tantalum Element (Information Table)
The important data related to tantalum element is given in the table below.
| Appearance of tantalum | Metallic gray with bluish tint |
| Atomic number of tantalum | 73 |
| Symbol of tantalum | Ta |
| Atomic mass of tantalum | 180.95 u |
| Protons, Neutrons & Electrons in tantalum | Protons: 73, Neutrons: 108, Electrons: 73 |
| State of tantalum (at STP) | Solid |
| Group number of tantalum in periodic table | 5 |
| Period number of tantalum in periodic table | 6 |
| Block of tantalum in periodic table | d-block |
| Category of tantalum | Transition metal |
| Bohr model or Electrons per shell or Electrons arrangement in tantalum | 2, 8, 18, 32, 11, 2 |
| Electron configuration of tantalum | [Xe] 4f14 5d3 6s2 |
| Orbital diagram of tantalum | 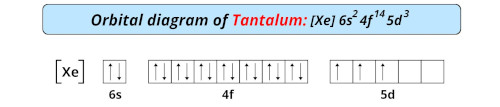 |
| Electronegativity of tantalum (on pauling scale) | 1.5 |
| Atomic radius of tantalum (van der Waals radius) | 217 picometers |
| Density of tantalum | 16.65 g/cm3 |
| 1st ionization energy of tantalum | 7.89 eV |
| Main isotope of tantalum | 181Ta |
| Melting point of tantalum | 3290 K or 3017 °C or 5463 °F |
| Boiling point of tantalum | 5731 K or 5458 °C or 9856 °F |
| Crystal structure of tantalum | Body Centered Cubic (BCC) |
| Discovery of tantalum | By Anders Gustaf Ekeberg in 1801 |
Also see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table).
Tantalum element in Periodic table
The Tantalum element (Ta) has the atomic number 73 and is located in group 5 and period 6. Tantalum is a metal and it is classified as a transition element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Click on above elements in the periodic table to see their information.
Facts about tantalum
Here are a few interesting facts about the tantalum element.
- Anders Gustaf Ekeberg discovered tantalum in the year 1802.
- Tantalum was given its name from the Greek mythological character “Tantalos”.
- Earth’s crust contains around 1.7 ppm (by weight) of tantalum.
- Australia produces the largest amount of tantalum than any other region.
- Most of the tantalum is obtained from the ore Columbite and Tantalite.
Properties of tantalum
Here is a list of some physical properties and chemical properties of tantalum.
Physical properties of tantalum
- Tantalum has a gray shiny metallic appearance.
- Tantalum can be drawn into thin wires because of its ductility.
- Tantalum can conduct heat and electricity.
- Tantalum has a BCC crystal structure.
- There are many isotopes of tantalum and out of those isotopes, the most abundant isotope is 181Ta.
- The melting point and boiling point of tantalum is 3290 K and 5731 K respectively.
Chemical properties of tantalum
- Tantalum is basically non reactive to most of the chemicals at room temperature.
- The oxide layer on the tantalum also protects it from further oxidation.
- Tantalum reacts with hydrofluoric acid at room temperature.
Uses of tantalum
Here are some uses of the tantalum element.
- Tantalum is used in capacitors, resistors, and other electrical devices.
- Tantalum oxide is used in manufacturing camera lenses that require a higher refractive index.
- Tantalum has a very high melting point and so it is added to other metals to increase the melting point.
- Tantalum is also used in some parts of missiles and aircrafts.
External resources:
- P. (n.d.). Titanium | Ti (Element) – PubChem. Titanium | Ti (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Titanium
- It’s Elemental – The Element Titanium. (n.d.). It’s Elemental – the Element Titanium. https://education.jlab.org/itselemental/ele022.html
- Titanium – Wikipedia. (2022, January 19). Titanium – Wikipedia. https://en.wikipedia.org/wiki/Titanium
- Titanium – Element information, properties and uses | Periodic Table. (n.d.). Titanium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/22/titanium
- Titanium. (n.d.). Titanium. https://webbook.nist.gov/cgi/inchi?ID=C7440326&Mask=20
- Haynes, W. M. (Ed.). (2014, June 4). CRC Handbook of Chemistry and Physics. https://doi.org/10.1201/b17118
- Kaye, G W.C., & Laby, T H. Tables of physical and chemical constants. 15th Edition. United States.
- Sansonetti, J. E., & Martin, W. C. (2005, December). Handbook of Basic Atomic Spectroscopic Data. Journal of Physical and Chemical Reference Data, 34(4), 1559–2259. https://doi.org/10.1063/1.1800011
- Bondi, A. (1964, March). van der Waals Volumes and Radii. The Journal of Physical Chemistry, 68(3), 441–451. https://doi.org/10.1021/j100785a001
- Holden, et al. (2018, December 1). IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Education Community (IUPAC Technical Report). Pure and Applied Chemistry, 90(12), 1833–2092. https://doi.org/10.1515/pac-2015-0703
- Zhang, Y., Evans, J. R. G., & Yang, S. (2011, January 11). Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. Journal of Chemical & Engineering Data, 56(2), 328–337. https://doi.org/10.1021/je1011086
- Possolo, A., van der Veen, A. M. H., Meija, J., & Hibbert, D. B. (2018, January 4). Interpreting and propagating the uncertainty of the standard atomic weights (IUPAC Technical Report). Pure and Applied Chemistry, 90(2), 395–424. https://doi.org/10.1515/pac-2016-0402
- Emsley, J. (2011). Nature’s Building Blocks: An A-Z Guide to the Elements. United Kingdom: OUP Oxford.
- Allred, A. (1961, June). Electronegativity values from thermochemical data. Journal of Inorganic and Nuclear Chemistry, 17(3–4), 215–221. https://doi.org/10.1016/0022-1902(61)80142-5
- James A. M. & Lord M. P. (1992). Macmillan’s chemical and physical data. Macmillan.
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
