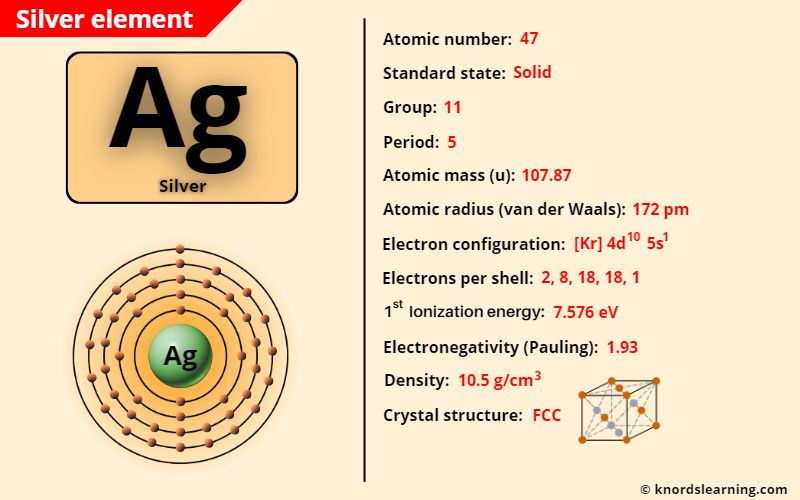
Silver element (Ag) is in group 11 and period 5 of a periodic table. Silver is in the d-block and it is classified as a transition element on the periodic table.
There is a lot more information related to silver which is mentioned in the Information Table given below.
So let’s dive right into it!
Table of contents
- Silver element (Information Table)
- Silver element in Periodic table
- Facts about Silver
- Properties of Silver
- Uses of Silver
Silver Element (Information Table)
The important data related to silver element is given in the table below.
| Appearance of silver | White metallic luster |
| Atomic number of silver | 47 |
| Symbol of silver | Ag |
| Atomic mass of silver | 107.87 u |
| Protons, Neutrons & Electrons in silver | Protons: 47, Neutrons: 61, Electrons: 47 |
| State of silver (at STP) | Solid |
| Group number of silver in periodic table | 11 |
| Period number of silver in periodic table | 5 |
| Block of silver in periodic table | d-block |
| Category of silver | Transition metal |
| Bohr model or Electrons per shell or Electrons arrangement in silver | 2, 8, 18, 18, 1 |
| Electron configuration of silver | [Kr] 4d10 5s1 |
| Orbital diagram of silver | 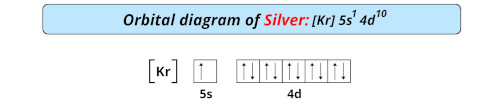 |
| Electronegativity of silver (on pauling scale) | 1.93 |
| Atomic radius of silver (van der Waals radius) | 172 picometers |
| Density of silver | 10.5 g/cm3 |
| 1st ionization energy of silver | 7.576 eV |
| Main isotope of silver | 107Ag (51.8%) and 109Ag (48.1%) |
| Melting point of silver | 1234.9 K or 961.7 °C or 1763.2 °F |
| Boiling point of silver | 2435 K or 2162 °C or 3924 °F |
| Crystal structure of silver | Face Centered Cubic (FCC) |
Also see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table).
Silver element in Periodic table
The Silver element (Ag) has the atomic number 47 and is located in group 11 and period 5. Silver is a metal and it is classified as a transition element.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Click on above elements in the periodic table to see their information.
Facts about silver
Here are a few interesting facts about the silver element.
- Silver was given its name from the Anglo-Saxon word “seolfor”.
- Silver was given more value than gold in ancient Egypt.
- Pound sterling is a currency of the UK and it was initially equal in value to 1 pound of silver.
- Silver is a ductile metal and it can be drawn into very thin wires.
- Mexico is the world’s largest producer of silver.
- Silver metal is generally obtained while mining copper, lead and zinc.
Properties of silver
Here is a list of some physical properties and chemical properties of silver.
Physical properties of silver
- Silver is a metal having a white shiny appearance.
- Silver metal is the best conductor of heat and electricity.
- The melting point and boiling point of silver metal is 1234.9 K and 2435 K respectively.
- Silver is ductile as well as malleable metal, that means it can be easily drawn into thin wires and sheets.
- Silver has many isotopes and out of these isotopes, the most abundant isotopes are 107Ag and 109Ag.
- The crystal structure of silver is FCC.
- Silver has an atomic mass of 107.87 u and its density is 10.5 g/cm3.
Chemical properties of silver
- The electron configuration of silver is [Kr] 4d10 5s1 and it is a transition metal.
- Silver gets a dark gray color if it is kept open in the air for a very long time. Because of this reason, it requires polishing.
- Silver has no reaction with water, acids as well as many other compounds.
- The black sulfide layer is formed on the silver metal when it is exposed to hydrogen sulfide (H2S).
Uses of silver
Here are some uses of the silver element.
- Silver is used in jewelry as it is a precious metal.
- Silver is also used in making components of batteries as well as in other electrical devices.
- Silver is also used in mirrors as it has an ability to reflect the light.
- During ancient times, the wines, vinegar, water, etc were stored in silver bottles to protect them from bacteria contamination.
- Silver metal was also used for filling cavities.
- Very thin layer of silver is applied on the sweets for decoration and we can also eat it.
External resources:
- Sansonetti, J. E., & Martin, W. C. (2005, December). Handbook of Basic Atomic Spectroscopic Data. Journal of Physical and Chemical Reference Data, 34(4), 1559–2259. https://doi.org/10.1063/1.1800011
- Bondi, A. (1964, March). van der Waals Volumes and Radii. The Journal of Physical Chemistry, 68(3), 441–451. https://doi.org/10.1021/j100785a001
- Holden, et al. (2018, December 1). IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Education Community (IUPAC Technical Report). Pure and Applied Chemistry, 90(12), 1833–2092. https://doi.org/10.1515/pac-2015-0703
- Silver – Element information, properties and uses | Periodic Table. (n.d.). Silver – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/47/silver
- Silver – Wikipedia. (2017, December 17). Silver – Wikipedia. https://en.wikipedia.org/wiki/Silver
- P. (n.d.). Silver | Ag (Element) – PubChem. Silver | Ag (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Silver
- It’s Elemental – The Element Silver. (n.d.). It’s Elemental – the Element Silver. https://education.jlab.org/itselemental/ele047.html
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/47.shtml
- Atomic Weight of Silver | Commission on Isotopic Abundances and Atomic Weights. (n.d.). Atomic Weight of Silver | Commission on Isotopic Abundances and Atomic Weights. https://ciaaw.org/silver.htm
- Atomic Data for Silver (Ag). (n.d.). Atomic Data for Silver (Ag). https://physics.nist.gov/PhysRefData/Handbook/Tables/silvertable1.htm
- Silver | Ag | ChemSpider. (n.d.). Silver | Ag | ChemSpider. http://www.chemspider.com/Chemical-Structure.22394.html?rid=9f8fd898-2e75-4ea6-8666-ed34595a2c3c
- Silver Statistics and Information | U.S. Geological Survey. (n.d.). Silver Statistics and Information | U.S. Geological Survey. https://www.usgs.gov/centers/national-minerals-information-center/silver-statistics-and-information
- C&EN: IT’S ELEMENTAL: THE PERIODIC TABLE – SILVER. (n.d.). C&EN: IT’S ELEMENTAL: THE PERIODIC TABLE – SILVER. https://pubsapp.acs.org/cen/80th/silver.html?
- Prohaska, T., et al. (2022, May 1). Standard atomic weights of the elements 2021 (IUPAC Technical Report). Pure and Applied Chemistry, 94(5), 573–600. https://doi.org/10.1515/pac-2019-0603
- Haynes, W. M. (Ed.). (2014, June 4). CRC Handbook of Chemistry and Physics. https://doi.org/10.1201/b17118
- Kaye, G W.C., & Laby, T H. Tables of physical and chemical constants. 15th Edition. United States.
- Zhang, et al. (2011, January 11). Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. Journal of Chemical & Engineering Data, 56(2), 328–337. https://doi.org/10.1021/je1011086
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
