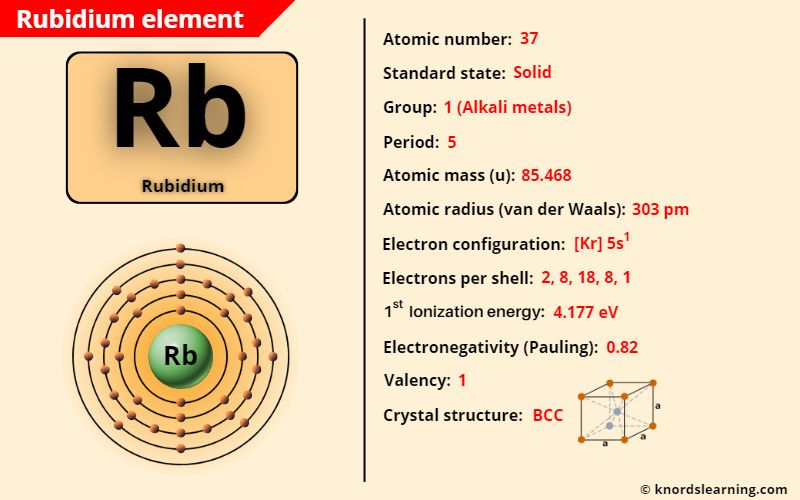
Rubidium element (Rb) is in group 1 and period 5 of a periodic table. Rubidium is in the s-block and it is classified as an alkali metal on the periodic table.
There is a lot more information related to rubidium which is mentioned in the Information Table given below.
So let’s dive right into it!
Table of contents
- Rubidium element (Information Table)
- Rubidium element in Periodic table
- Facts about Rubidium
- Properties of Rubidium
- Uses of Rubidium
Rubidium Element (Information Table)
The important data related to rubidium element is given in the table below.
| Appearance of rubidium | Grey white |
| Atomic number of rubidium | 37 |
| Symbol of rubidium | Rb |
| Atomic mass of rubidium | 85.468 u |
| Protons, Neutrons & Electrons in rubidium | Protons: 37, Neutrons: 48, Electrons: 37 |
| State of rubidium (at STP) | Solid |
| Group number of rubidium in periodic table | 1 |
| Period number of rubidium in periodic table | 5 |
| Block of rubidium in periodic table | s-block |
| Category of rubidium | Alkali metals |
| Bohr model or Electrons per shell or Electrons arrangement in rubidium | 2, 8, 18, 8, 1 |
| Electron configuration of rubidium | [Kr] 5s1 |
| Orbital diagram of rubidium |  |
| Valence electrons in rubidium | 1 |
| Electronegativity of rubidium (on pauling scale) | 0.82 |
| Atomic radius of rubidium (van der Waals radius) | 303 picometers |
| Density of rubidium | 1.53 g/cm3 |
| 1st ionization energy of rubidium | 4.177 eV |
| Main isotope of rubidium | 85Rb, 87Rb |
| Melting point of rubidium | 312.4 K or 39.3 °C or 102.7 °F |
| Boiling point of rubidium | 961 K or 688 °C or 1270 °F |
| Crystal structure of rubidium | Body Centered Cubic (BCC) |
| Discovery of rubidium | By Robert Bunsen and Gustav Kirchhoff (in 1861) |
Also see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table).
Rubidium element in Periodic table
The Rubidium element (Ru) has the atomic number 37 and is located in group 1 and period 5. Rubidium is in solid state at STP and it is classified as an alkali metal on the periodic table.
| H | He | ||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Ru | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La* | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| Fr | Ra | Ac** | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| *Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ||||
| **Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr |
Click on above elements in the periodic table to see their information.
Facts about rubidium
Here are a few interesting facts about rubidium element.
- The name “Rubidium” came from the Latin word “rubidus” (which means deep red).
- Rubidium is the second most electropositive element.
- Rubidium is the 23rd most abundant element found from the earth’s crust.
- The amount of rubidium in the seawater is approximately 125 µg/L.
- The density of rubidium is 1.5 times the density of water.
Properties of rubidium
Here is a list of some physical properties and chemical properties of rubidium.
Physical properties of rubidium
- Rubidium is a soft metal and it has a gray-white appearance.
- The melting point of rubidium is 39.3 °C, which is slightly higher than our body temperature (our body temperature is 37 °C).
- The boiling point of rubidium is 688 °C.
- Rubidium has many isotopes, but the stable isotopes that are found from the earth’s crust are 85Rb and 87Rb.
Chemical properties of rubidium
- Rubidium is a reactive metal and it is not found in its pure form. It is always found as a compound with other elements in the earth’s crust.
- The rubidium metal gives a red-violet color in flame test.
- Rubidium reacts with oxygen to form a number of oxides (like Rb2O, Rb6O, Rb9O2, etc).
- Rubidium metal reacts violently with water and releases heat. This heat can even ignite the hydrogen gas which is released during the reaction.
Uses of rubidium
Here are some uses of the rubidium element.
- The compounds of rubidium are used in fireworks to produce the red-violet color.
- Rubidium is a highly reactive metal and hence it is mostly used in research work in chemistry.
External resources:
- It’s Elemental – The Element Rubidium. (n.d.). It’s Elemental – the Element Rubidium. https://education.jlab.org/itselemental/ele037.html
- Georgescu, I. (2015, November 20). Rubidium round-the-clock. Nature Chemistry, 7(12), 1034–1034. https://doi.org/10.1038/nchem.2407
- Simmons, E. C. (n.d.). Rubidium: Element and geochemistry. Encyclopedia of Earth Science, 555–556. https://doi.org/10.1007/1-4020-4496-8_278
- Possolo, et al. (2018, January 4). Interpreting and propagating the uncertainty of the standard atomic weights (IUPAC Technical Report). Pure and Applied Chemistry, 90(2), 395–424. https://doi.org/10.1515/pac-2016-0402
- Emsley, J. (2011). Nature’s Building Blocks: An A-Z Guide to the Elements. United Kingdom: OUP Oxford.
- Haynes, W. M. (Ed.). (2014, June 4). CRC Handbook of Chemistry and Physics. https://doi.org/10.1201/b17118
- Electronic structure of the elements. (2000, March). The European Physical Journal C, 15(1–4), 78–79. https://doi.org/10.1007/bf02683401
- Rubidium – Element information, properties and uses | Periodic Table. (n.d.). Rubidium – Element Information, Properties and Uses | Periodic Table. https://www.rsc.org/periodic-table/element/37/rubidium
- P. (n.d.). Rubidium | Rb (Element) – PubChem. Rubidium | Rb (Element) – PubChem. https://pubchem.ncbi.nlm.nih.gov/element/Rubidium
- It’s Elemental – The Element Rubidium. (n.d.). It’s Elemental – the Element Rubidium. https://education.jlab.org/itselemental/ele037.html
- James A. M. & Lord M. P. (1992). Macmillan’s chemical and physical data. Macmillan.
- Bedford, et al. (1996, April 1). Recommended values of temperature on the International Temperature Scale of 1990 for a selected set of secondary reference points. Metrologia, 33(2), 133–154. https://doi.org/10.1088/0026-1394/33/2/3
- Allred, A. (1961, June). Electronegativity values from thermochemical data. Journal of Inorganic and Nuclear Chemistry, 17(3–4), 215–221. https://doi.org/10.1016/0022-1902(61)80142-5
- Periodic Table of Elements: Los Alamos National Laboratory. (n.d.). Periodic Table of Elements: Los Alamos National Laboratory. https://periodic.lanl.gov/37.shtml
- Cesium and Rubidium Statistics and Information | U.S. Geological Survey. (n.d.). Cesium and Rubidium Statistics and Information | U.S. Geological Survey. https://www.usgs.gov/centers/national-minerals-information-center/cesium-and-rubidium-statistics-and-information
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. With a desire to make learning accessible for everyone, he founded Knords Learning, an online learning platform that provides students with easily understandable explanations.
Read more about our Editorial process.
